U.S. Navy Aeromedical Reference and Waiver Guide Medications - 1
18.0 MEDICATIONS
Last Reviewed: March 2022 Last Revised: March 2022
Note: Any medication not listed in this section is not approved for aviation. Contact NAMI
Code 53HN if further guidance is needed.
18.1 NATOPS ON MEDICATIONS
General NATOPS (OPNAVINST 3710.7 series, chapter 8) includes the following statements on
medications (Drugs):
Taking drugs prescribed by competent medical authority shall be considered sufficient cause for
recommendation of grounding unless their use is specifically approved by a Flight Surgeon (or
Aeromedical Examiner or Aeromedical Physician Assistant), or a waiver for specific drug use
has been granted by CHNAVPERS or the Commandant of the Marine Corps. Consideration
shall be given to the removal of ground support personnel from critical duties, for the duration of
the drug effects, if appropriate. Medications such as antihistamines, antibiotics, tranquilizers,
sleeping pills, etc., shall be discarded if all are not used during the period of medication.
Because of the possibility of adverse side effects and unpredictable reactions, the use of over-
the-counter drugs by flight personnel is prohibited unless specifically approved by a Flight
Surgeon (or Aeromedical Examiner or Aeromedical Physician Assistant). Ground support
personnel shall be briefed on the hazards of self-medication and should be discouraged from
using such drugs.
In general, all medications require temporary grounding unless specifically described here as
NCD for flight duties.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 2
18.2 ANTIMICROBIAL
Last Reviewed: August 2019 Last Revised: August 2019
All antibiotics other than the following very specific exceptions require grounding (CD). The
listed exceptions do not forgive you from doing something obviously inadvisable such as
allowing a sick person to fly.
Aviation personnel on the following approved antibiotics may be considered for an up chit prior
to the completion of the course of therapy as long as the condition being treated has resolved in
all significant aspects with no adverse reaction that might compromise safety of flight or mission
completion.
ANTI-BACTERIAL MEDICATIONS:
ANTI-MALARIALS:
Refer to Aeromedical Reference and Waiver Guide (ARWG) section 17 on Malaria.
FLUOROQUINOLONES:
CIPROFLOXACIN: NCD.
ISONIAZID: No waiver needed when used for TB prophylaxis as long as the member
remains under close evaluation by flight surgeon. This medication causes occasional liver
damage, especially above age 35. All personnel are to be monitored in accordance with current
preventive and occupational medicine guidelines.
MACROLIDES:
ERYTHROMYCIN: NCD- including long-term, low-dose use for acne.
NITROFURANTOIN: CD. Waiver considered if under close observation of flight surgeon.
Adverse effects include pneumonitis or peripheral neuropathy.
PENICILLINS:
AMPICILLIN, AMOXICILLIN, PENICILLIN VK, AUGMENTIN, DICLOXACILLIN: NCD.
SULFONAMIDES:
BACTRIM/SEPTRA: CD. Waivers will be considered for long term use.
TETRACYCLINES:
TETRACYCLINE, DOXYCYCLINE: NCD. (Including long-term use for acne).
MINOCYCLINE: CD. Prohibited due to possible vestibular side effects.
ANTI- FUNGAL MEDICATIONS:
GRISEOFULVIN: CD. Waivers are considered if under close observation by local flight
surgeon. Watch for bone marrow suppression.
ITRACONAZOLE (SPORANOX): NCD. While not approved for chronic use,
itraconazole has a safer profile than ketoconazole, and need not be used on a chronic basis to
be effective. Recommended use in aviation personnel is to administer in week-long pulses each
month for four to six cycles. Aviators should be grounded for the first 48 hours of each cycle.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 3
Since it is not administered chronically, such as griseofulvin, a waiver is not required. The
recommended initial treatment is over a weekend to allow return to flight duties the following
Monday, thus minimizing flight schedule loss.
TERBINAFINE (LAMISIL): NCD. Requires a 72-hour grounding period. Terbinafine
has a safer profile than ketoconazole and has a lower relapse rate than itraconazole. The
recommended use in aviation personnel is to administer daily for twelve weeks. Aviators should
be grounded for the first 72 hours and a waiver is not required when no side-effects exist and
appropriate monitoring is performed. The recommended initial treatment is over a weekend to
allow return to flight duties the following Monday, thus minimizing flight schedule loss.
ANTI-VIRAL MEDICATIONS:
ACYCLOVIR, VALACYCLOVIR: NCD for intermittent and continuous/suppressive
therapy. The patient should be grounded and monitored for side effects for a minimum of 3
days during the initial treatment or upon initiation or re-initiation of suppressive therapy. If these
medications are being used to treat an active outbreak of herpes simplex or herpes zoster, the
flight surgeon should give consideration to the impact of active lesions on safety of flight,
especially the impact on safe of use of life support equipment (mask, harness, etc.). Should
there be concern for safety of flight, a grounding period is warranted, even though the disease
itself does not require a waiver. The need for suppressive therapy should be reassessed on an
annual basis. Topical acyclovir is also NCD. If a member is currently on a waiver for use, only
submit for waiver to be vacated at next required routine submission with an AERO Generated
AMS.
OSELTAMIVIR (TAMIFLU), ZANAMIVIR (RELENZA)- NCD, Requires a 72-hour
grounding period. These medications are indicated for prophylaxis and treatment of influenza
A and B viruses. They can decrease the severity, duration and complications of influenza
illnesses. These medications require a 72-hour grounding period following initiation of treatment
to assess for adverse side effects. In the absence of flu symptoms and adverse side effects
from the medications, flight duties may resume following the 72 hour grounding period.
Reducing the initial grounding period to 48 hours may be considered for operational
requirements with NAMI consultation.
TRUVADA® (EMTRICITABINE + TENOFOVIR) PRE-EXPOSURE PROPHYLAXIS:
CD, waivers considered for specific use on a case-by-case basis. See entry in
Miscellaneous section for details.
U.S. Navy Aeromedical Reference and Waiver Guide Medications - 4
18.3 ANTI-HYPERLIPIDEMICS
EZETIMIBE (ZETIA): NCD. A waiver is not required. An initial grounding period for 72
hours is required to assess for idiosyncratic reactions. If used in combination with HMG-CoA
reductase inhibitors, refer to the waiver guide section on Hyperlipidemia for additional guidance.
FIBRIC ACIDS:
FENOFIBRATE (TRICOR); GEMFIBROZIL (LOPID): CD. Fenofibrate (Tricor) and
gemfibrozil (Lopid) are both considered disqualifying. A waiver may be considered after a 14-
day ground trial of the medication without side-effects. Fenofibrate is preferred over gemfibrozil
due to fewer side effects. Prior to initiating treatment, baseline lab studies must be obtained to
include: lipid panel, liver function testing (ALT/AST/ALK PHOS), CBC, FBS, and CPK. These
tests are to be repeated at three months, six months and then annually if the values remain
stable. Evaluate for muscle aches (myalgias) at follow-up exams and measure CPK levels if
clinically indicated. If fibric acid is used in combination with an HMG-CoA reductase inhibitor,
refer to the waiver guide section on Hyperlipidemia for further guidance.
NIACIN: CD. No waiver.
RESINS:
CHOLESTYRAMINE: NCD if tolerated without side effects.
STATINS:
PRAVASTATIN, SIMVASTATIN, LOVASTATIN, ATORVASTATIN: NCD. HMG Co-A
reductase inhibitors (pravastatin, simvastatin, lovastatin, atorvastatin, etc.) are all NCD and a
waiver is not required. Refer to ARWG section on hypercholesterolemia for additional guidance.
Lipid panel, liver function tests (ALT/AST/ALK PHOS), CBC, and CPK are recommended at
baseline, 3, and 6 months, then annually. Liver enzyme elevations above three times normal are
disqualifying.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 5
18.4 ANTI-HYPERTENSIVES
Last Reviewed: December 15 Last Revised: October 15
ACE INHIBITORS (ACE-I):
CD. The entire family (captopril, enalapril, lisinopril, etc.) is CD, but waiverable. The
member must be grounded upon initiation of treatment. Waiver will be considered after 30 days
of treatment if member's hypertension is controlled on a stable dosage of medication without
evidence of side effects. If local pharmacy policy requires changing from one ACE-I to another,
advise Code 53HN of the change. Refer to ARWG section on hypertension for additional
guidance.
ANGIOTENSIN RECEPTOR BLOCKERS (ARB):
CD. These agents may be used if member does not tolerate an ACE-I or has some other
specific medical indication for its use. The same guidelines used for ACE-I apply.
ANTIADRENERGIC AGENTS:
DOXAZOSIN, PRAZOSIN, others in class: CD. No Waiver. Call NAMI Code 53HN for
further guidance.
BETA BLOCKERS (for hypertension only):
CD. Beta blockers are not considered for waivers for Service Groups I or II personnel.
Senior officers (LCDR and above) may be waived to Service Group 3 or Class II flying duties in
non-tactical aircraft. All SG 1, SG 2, or tactical NFOs are considered NPQ, no waiver
recommended. Designated Naval Aircrew will be considered for a waiver. Aviation personnel on
beta blockers should not pull more than 2.5 Gs, so requests for waivers should state
"transport/maritime/helo aircraft only." Air traffic controllers are usually waived. When beta
blockers are used, preference shall be given to cardioselective agents such as atenolol.
CALCIUM CHANNEL BLOCKERS:
AMLODIPINE (NORVASC): CD. A second generation calcium channel blocker,
AMLODIPINE may be considered for waiver for use in the control of hypertension only after
failure to control the condition on other approved agents. These cases must be reviewed
individually by NAMI prior to issuance of an Aeromedical Clearance notice. Local Board is not
authorized to issue a clearance notice for AMLODIPINE use.
NIFEDIPINE (PROCARDIA): CD. No Waiver.
COMBINATION AGENTS:
CD. Combination agents may be used if the individual agents themselves are
recommended for waiver. Follow the restrictions and guidelines outlined for each individual
agent.
THIAZIDE DIURETICS:
HYDROCHLOROTHIAZIDE (for Hypertension): CD. HYDROCHLOROTHIAZIDE
(HCTZ), with or without triamterene or potassium replacement, can be used as a first line agent
for treatment of hypertension in designated personnel. ACE inhibitors are preferred as they
have a low incidence of aeromedically significant side effects and are generally well tolerated.
See hypertension section of ARWG for waiver criteria and further guidance.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 6
18.5 IMMUNIZATIONS
Last Reviewed: August 2021 Last Revised: August 2021
GROUNDING FOR VACCINATIONS:
OPNAVINST 3710.7 series requires a 12 hour grounding period following immunizations unless
otherwise specified in this document. The specific guidelines and grounding periods for each
vaccination are described below. As per MANMED Article 15-77, the administration of routine
immunizations that require a temporary grounding, do not require issuance of an Aeromedical
Grounding Notice. This is a “self-limited” grounding period allowed in the absence of adverse
side effects.
VACCINE ADVERSE EVENT REPORTING SYSTEM (VAERS)
The Vaccine Adverse Event Reporting System (VAERS) is used to report adverse events or
reactions to all vaccines. VAERS, the primary U.S. vaccine safety monitoring system,
encourages reporting of any unexpected or serious event occurring after any vaccination as well
as adverse events occurring in persons following close contact with a vaccine recipient. An
adverse event is any clinically significant medical event that occurs following administration of a
vaccine. A VAERS report should be submitted even if it is not certain that the event was caused
by the vaccine. Web reporting is available at http://vaers.hhs.gov/.
ANTHRAX
BACKGROUND: Human anthrax vaccine was developed in England and the U.S. in the 1950s
and early 1960s. The vaccine is U.S. Food and Drug Administration (FDA)-licensed and has
been routinely given in the U.S. since 1970.
The vaccine has an excellent safety record. The most common side effects reported are mild
discomfort (localized swelling and redness at the site of injection), joint aches, and in a few
cases, nausea, loss of appetite, and headaches. There is no evidence from records at the
Michigan Biologic Products Institute (which is the only U.S. producer of the vaccine) that the
vaccine is associated with permanent local or systemic effects.
DOSAGE AND ADMINISTRATION: The current dose schedule for the U.S. vaccine consists of
6 shots given over an 18 month schedule and an annual booster thereafter.
Contraindications for use are sensitivity to vaccine components (formalin, aluminum hydroxide,
benzethonium chloride) and/or history of clinical anthrax. Pregnant women should not receive
this vaccine until after delivery. The vaccine should be stored at refrigerator temperature (not
frozen).
A 12 hour grounding period is recommended for the anthrax vaccination.
CHOLERA
Sale of the only licensed cholera vaccine in the United States has been discontinued, and the
CDC does not currently recommend the vaccine for travelers because of the brief and
incomplete immunity it offers. In lieu of vaccination, proper hygiene and food and water
precautions should be carefully emphasized.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 7
DIPHTHERIA TETANUS (DT) AND TETANUS TOXOID
This vaccine is used to prevent bacterial elaboration of toxins resulting in muscular
spasm/lockjaw, which is usually found in the setting of a contaminated wound. These vaccines
are toxoids and are both known to be 95% efficacious. They are given every 10 years, however
if a suspicious wound is encountered, the standard is to revaccinate if more than 5 years has
elapsed since the last vaccination. The dose is 0.5 cc IM. Adverse events include frequent local
reactions. Hypotonic, hyporesponsive episodes, seizures, and acute encephalopathy have been
reported on rare occasions. A 12 hour grounding period is recommended for this vaccination.
HEPATITIS A
This is an inactivated virus vaccine which is given as a 1.0 cc dose IM, with a booster dose 6 to
12 months later. Protective levels of antibodies are detectable in 80 to 98% of recipients 15
days after the first dose, and in 96% after one month. Protection is expected to last 20 years. No
significant adverse events have been reported, although some recipients experience local
injection site soreness. Transient systemic symptoms are uncommon. In the USA, the presence
of anti-HAV antibodies indicating past infection and probable immunity increases from about
10% in young children to about 75% in adults more than 50 years old. A 12 hour grounding
period is recommended for this vaccination.
HEPATITIS B
This is an inactivated virus vaccine which is given as a 1.0 cc IM dose, with boosters at one and
6 months. Current CDC recommendations are to immunize everyone 18 years of age or
younger and adults over 18 who are at risk. The at-risk population includes health care and
public safety worker who might have contact with blood or body fluids, people who have more
than one sex partner in six months, sex contacts of infected people, people who inject illegal
drugs, hemodialysis patients, and household contacts of people with chronic HBV infection.
Contraindications to vaccination include a history of allergic reaction to either baker's yeast or
the hepatitis B vaccine. Mild soreness at the injection site is seen in approximately one out of 11
children and adolescents and one out of four adults, and mild to moderate fever is seen in up to
one out of 14 children and one out of 100 adults. A 12 hour grounding period is recommended
for this vaccination.
INFLUENZA
INJECTABLE INACTIVATED INFLUENZA VACCINE
This vaccine is composed of inactivated whole or disrupted influenza viruses and changed
annually to reflect antigenic changes in the A and B strains of the virus that is in circulation.
Immunity after the standard 0.5 cc IM dose lasts about six months, so annual administration is
required, ideally before the start of flu season. The vaccine is indicated in the elderly (>65),
residents of chronic care facilities, those with cardiac, pulmonary or immunosuppressive
diseases such as cancer and DM, and close contacts of those at risk. All active duty Navy and
Marine Corps personnel are required to have one dose of this vaccine each year. The only
contraindication is a bona fide history of generalized allergic reaction to the vaccine, eggs, or
egg components. Effectiveness varies with how closely vaccine strains match the strains in the
community, generally about 60-85%. A mild local reaction is the most common adverse effect,

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 8
although some individuals have a transient mild "viral syndrome." A 12 hour grounding period is
recommended for this vaccination.
FLUMIST
All active duty Navy and Marine Corps personnel are required to have one dose of influenza
vaccine (IM or intranasal spray) each year. FluMist® (Influenza Virus Vaccine Live, Intranasal),
is composed of live, attenuated influenza virus (LAIV) that is administered by nasal spray. It is
used for the prevention of Influenza A and B in healthy adults under age 50 who are not
pregnant. The 0.5mL dose is given as a 0.25mL spray in each nostril.
The immunization is less effective in those with pre-existing nasal congestion. The dose should
be repeated if the patient sneezes following administration. Immunity after the standard
intranasal dose declines during the year, so annual administration is required—ideally, before
the start of “flu season.” There appears to be an increase in protective antibodies over time with
subsequent doses. Effectiveness varies according to how closely the strains used to make the
vaccine match those in the community.
The onset of symptoms after immunization usually occurs within the first 24 hours, with most
symptoms presenting by the third day. The duration of symptoms is typically 1-2 days. The most
common adverse effects include:
• headache 40%
• sore throat 28%
• tiredness 26%
• myalgias 17%
• cough, nasal congestion, and rhinitis 9-45%
• Less common adverse effects include chills, abdominal pain, diarrhea, vomiting, and
otitis media.
A “self-limited” grounding period of 72 hours after immunization is required to assess for
symptom severity. Commanding officers may return aeronautically designated personnel to duty
involving flight operations in less than 72 hours on the recommendation of a flight surgeon when
necessary to meet “real world” operational commitments. The presence and severity of
symptoms may require the grounding of some personnel for greater than 72 hours. To minimize
operational impact, commands may elect to stagger the administration of the vaccine to their
personnel. For example, a command might elect to vaccinate 50% of eligible personnel one
week and the remaining personnel the following week. Another option would be to schedule
immunizations immediately prior to a period when no flights are scheduled (e.g., just prior to a
holiday weekend).
Additional information is available via the CDC website at,
http://www.cdc.gov/flu/professionals/vaccination/
ANTIVIRAL MEDICATIONS:
OSELTAMIVIR (TAMIFLU), ZANAMIVIR (RELENZA)-
NCD, Requires an initial 72-hour grounding period. See Antimicrobial Section 18.2.
JAPANESE ENCEPHALITIS (March 2010)
Japanese Encephalitis (JE), a mosquito-borne arboviral infection, is the leading cause of viral
encephalitis in Asia with over 50,000 sporadic and epidemic cases reported annually. Two

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 9
inactivated virus vaccines are currently available, JE-Vax, licensed in 1993, and Ixiaro, licensed
in 2009.
JE-VAX. JE-Vax is administered as a 1.0 mL SC dose with an effectiveness of 80-90%.
Intradermal dosing at two sites is as immunogenic as a single SC dose. Three doses during a
30 day period (days 0, 7, and 30) provides the longest immunogenic protection. A booster given
at one year will significantly increase antibody titers, which may then persist for several years.
An abbreviated schedule of immunizations given on days 0, 7, and 14 may be used if significant
time constraints exist.
JE-Vax is associated with a moderate frequency of local and mild systemic side effects. About
20% of recipients experience local redness, swelling, or tenderness, and systemic side effects
(fever, headache, malaise, and rash) have been reported in about 10% of vaccine recipients. An
additional pattern of adverse reactions characterized by generalized urticaria and/or
angioedema, and rarely respiratory distress or collapse, has been reported. These reactions
occurred after a longer interval and usually after the first or second dose. The median time to
onset of symptoms after the first dose is 12 hours, and 88% of reactions occur within 3 days.
The interval after the second dose is longer, with a median time of 3 days and possibly as long
as two weeks. After reviewing the experiences of I-MEF personnel during the first several years
of use, the original 3-5-3 day grounding regimen appears excessive based upon the actual
observed incidence of reactions. A 24 hour grounding period is recommended after each
dose providing that aviators are formally briefed about possible delayed reactions.
Individuals who have a past history of urticaria or hypersensitivity phenomena should remain
under the previous guidelines (3-5-3 grounding).
JE-Vax is no longer produced. The DoD stockpile is projected to be exhausted in April 2010 and
has an expiration date of May 2011. Ixiaro is an available alternative to JE-Vax.
Ixiaro. Ixiaro is administered as a 0.5 mL IM dose. Two doses are given 28 days apart (days 0
and 28). The protective antibody response is 95% at six months and 83.4% at 12 months.
Ixiaro is associated with a moderate frequency of mild systemic and local side effects. About
20% of recipients experience headache, 15% experience myalgia, and 50% experience mild
local reactions in rates comparable to placebo. A 12 hour grounding period is recommended
for this vaccination.
Summary- Either JE-Vax or Ixiaro can be used for aviators as described above. JE-Vax will
become unavailable by May 2011 unless a shelf-life extension is approved. Ixiaro is likely to
have less serious adverse events than JE-Vax. Ixiaro requires only two doses, and requires a
12-hour grounding period instead of 24-hours for JE-Vax. Ixiaro is the preferred vaccine for
aviation, especially when the duration of the grounding period impacts mission accomplishment.
MEASLES/MUMPS/RUBELLA (MMR)
This vaccine, composed of live, attenuated viruses, is indicated in adults born after 1956 without
a history of documented measles or measles/mumps vaccination. Some people vaccinated
before 1980, especially if before 14 months of age, may be inadequately protected and now
require revaccination. Contraindications include pregnancy, immunosuppression (except HIV),
recent IG administration, or anaphylactic reactions to the immunization, eggs, or neomycin.
Efficacy is 95% for all three components. Serious adverse events are rare, but include acute
encephalopathy, parotiditis, and orchitis. Transient arthralgias may occur in up to half of first-
time recipients, but arthritis and arthropathy are rare. About 5-15% of vaccine recipients have

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 10
fever up to 21 days post-vaccination and 5% may develop a rash. One study assessed the
incidence of adverse events after revaccination. This study noted local injection site discomfort
and flu-like symptoms amongst 6.6% and 3.4% of male and female students respectively. The
4% rate of joint related complaints after revaccination was less than that found after primary
vaccination. A 12 hour grounding period is recommended for this vaccination.
MENINGOCOCCAL
Each year, approximately 2,600 people contract meningococcal disease. Of these, 10 to 15%
die. Of those who live, another 11 to 19% lose their arms or legs, become deaf, have problems
with their nervous system, become mentally retarded, or suffer from seizures or strokes. The
meningococcal vaccine is a polysaccharide vaccine that can prevent 4 types of meningococcal
disease including 2 of the 3 most common in the United States and a type that causes
epidemics in Africa. It is administered as a 0.5 cc SC dose, and is recommended for all children
at their preadolescent visit, military recruits, college freshman living in dormitories,
microbiologists who might be exposed to the bacteria, anyone with an immune system disorder,
asplenic patients, people who might have been exposed to meningitis during an outbreak, and
anyone traveling to or living in a part of the world where meningococcal disease is common.
Approximately half of vaccine recipients experience mild side effects, such as pain or redness at
the injection site. A small percentage of patients also develop fever. Although rare, serious
allergic reactions can develop within a few minutes to hours of vaccination. Of note, a few cases
of Guillain-Barre syndrome have been reported among people who received the MCV4 vaccine,
however there is currently not enough information to determine if this was caused by the
vaccine. A 12 hour grounding period is recommended for this vaccination.
PLAGUE
This vaccine is composed of a suspension of killed bacteria, and is given as a dose of 1.0 cc IM.
It is used in laboratory workers and travelers to endemic areas. The vaccine is given as a series
with a primary dose as above, then 0.2 cc IM doses at 4 weeks and 6 months. Boosters are
given every 6 to 12 months as long as exposure continues. There is a 90 to 93% antibody
response however efficacy is uncertain. Up to 10% of recipients will develop local reactions.
Sterile abscesses and hypersensitivities have also been reported.
PNEUMOVAX (PPV23)
This vaccine was designed to decrease the risk of pneumococcal infection in susceptible
individuals such as military recruits, asplenic patients, immunosuppressed individuals, and
those over 65. This preparation consists of purified polysaccharide coats of 23 serotypes and is
considered to be 60 to 80% efficacious, reducing serious sequelae of infection by about 50%. In
asplenic patients it is about 13 -33% effective in producing a two-fold increase in antibody titer.
The dose is 0.5 cc IM or SC, and a booster is recommended in high-risk (transplant, nephrotic
syndrome, asplenic) individuals at 6 years. Pneumovax has been associated with a 50% local
reaction rate, an arthus-like reaction with booster doses, and rarely, anaphylaxis. A 12 hour
grounding period is recommended for this vaccination.
POLIO
The inactivated polio virus (IPV) is given as a dose of 0.5 cc IM or SC. The use of oral polio
vaccine (OPV) is no longer recommended. Travelers to endemic areas who have received
primary immunization during childhood should consider a single booster (IPV) in adulthood,

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 11
while those who were never vaccinated should be vaccinated according to current CDC
guidelines. A 12 hour grounding period is recommended for this vaccination.
SARS-COV-2 (COVID-19)
COVID-19 infection is caused by a novel coronavirus, SARS-CoV-2, and can lead to a spectrum
of symptoms and physical manifestations ranging from a mild fever and cough to significant
cardiopulmonary compromise. Infection with SARS-CoV-2 can adversely impact Navy and
Marine Corps force readiness and mission execution. Several SARS-CoV-2 vaccines of varying
platforms have been released under Federal Drug Administration (FDA) Emergency Use
Authorizations (EUA). Some vaccines utilize mRNA delivered via a lipid nanoparticle (LNP)
delivery system while others utilize a live-non-replicating viral vector.
The initial two SARS-CoV-2 vaccines are two-dose series with each dosing separated by 21-28
days. The first one-dose SARS-CoV-2 vaccine received FDA EUA in February of 2021. Navy
and Marine Corps personnel in a flight duty status may volunteer for administration of any
SARS-CoV-2 vaccine unless OPNAV or other operational directives provide restriction. Once a
vaccine series is initiated, the Service member should complete the series from the same
manufacturer and vaccine platform type. Service members should not be given a SARS-CoV-2
vaccine from different manufacturers. Receipt of a single dose of a two-dose mRNA vaccine
series does not contraindicate receipt of the Janssen vaccine for individuals who are unable to
complete the two-dose mRNA vaccine series and who were administered the first dose at least
28 days prior. This should be very infrequent and must be reported to the Vaccine Adverse
Event Reporting System (VAERS). Although serious side effects are rare, they can last for a
prolonged period of time, necessitating an extended grounding period. In order to minimize
impact on training and operations, personnel in a flight duty status should consult with their
Aerospace Medicine provider to determine the optimal timing to receive the vaccine.
Additionally, to minimize operational impact, commands may elect to stagger the administration
of the vaccine to their personnel. Personnel in a Class 1 or 2 flight duty status who have
received any dose of a SARS-CoV-2 vaccine course, and develop symptoms consistent with
COVID-19 outside of expected side effects, and before the projected window of final dose
immunity, should be tested for COVID-19. If the test result is positive and medical evaluation
results in a diagnosis of COVID-19, personnel and Aerospace medicine providers shall follow
ALNAV 096/20.
Nucleic Acid Vaccines
DNA Vaccines: No current recommendation at this time, as this type of vaccine has yet to
receive EUA from the FDA. Until a recommendation is published, personnel in a flight duty
status should not receive this vaccine platform.
Messenger RNA Vaccines: The Pfizer-BioNTech and Moderna SARS-CoV-2 vaccines are
variations of a messenger RNA (mRNA) platform. The vaccines are administered as a two-dose
intramuscular injection series. Both vaccines may cause side effects (SEs), which are defined
as reported symptoms shown to be linked to a vaccine by scientific studies. SE symptoms may
be local, such as pain, redness, and swelling at the injection site, or systemic, including fatigue,
headache, myalgias, nausea, and chills. During the Phase 3 randomized and placebo-controlled
trials of the Pfizer-BioNTech vaccine, over 70% of participants over the age of 16 experienced
local SEs after any dose, approximately 60% experienced systemic SE after the first dose, and
approximately 70% experienced systemic SE after the second dose (Pfizer-BioNTech, 2020).
The incidence of severe local reactions varied between 0-1%, and the incidence of severe
systemic SE varied between 0-4.6%. The median onset of local SE was 0-2 days with a median
duration of 1-2 days, and the median onset of systemic SE was 1-2 days with a median duration

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 12
of 1 day. During the Phase 3 randomized and placebo-controlled trials of the Moderna vaccine,
a majority of participants ages 18-63 reported local SE; 87% after dose one and 90% after dose
two. Systemic SEs were reported by 55% of participants after dose one and 79% of participants
after dose two (Moderna, 2021). Of these SEs, between four and seven percent of the local
reactions were rated as severe and between three and sixteen percent of the systemic reactions
were rated as severe. The median onset of both local and systemic SEs was one day with a
median duration of 2-3 days. The most common reported local SE was pain, and the most
common reported systemic SEs were headache and fatigue. Rare cases of myocarditis, most
commonly affecting young men, have also been reported following administration of the mRNA
vaccine platform, but a causal link has not been established.
If personnel in a flight duty status (all classes) receive a dose of a SARS-CoV-2 mRNA vaccine,
a “self-limited” grounding period of 48-hours is required to assess for onset of systemic SEs
(fatigue, headache, myalgia, nausea, fever, and chest pain) after any dose in the series. The
development or presence of any systemic SEs during these 48-hours requires extending the
“self-limited” grounding for a total of 96-hours, regardless of when the SEs resolve, to allow for
full recovery. If systemic SEs persist for greater than 96-hours, any personnel in a flight duty
status should see their Aerospace Medicine provider for evaluation. If any health problems
develop besides known side effects, personnel must also see their Aerospace Medicine
provider for evaluation. The presence and severity of symptoms may require the grounding of
some personnel for greater than 96-hours. Medical evaluations for return to flight status in these
prolonged symptomatic cases shall follow the return to flight status symptomatic protocols
outlined in ALNAV 096/20. Due to variations in vaccine composition between manufacturers,
timing of SARS-CoV-2 vaccination with other vaccinations should follow current Centers for
Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices’ (ACIP)
guidelines (Cohn and Mbaeyi, 2020). Aerospace Medicine providers administering the vaccine
should review further information provided by the FDA (U.S. Food & Drug Administration, 2021).
Live Non-replicating Viral Vector Vaccines
Replication-incompetent Adenovirus Type 26 (Ad26) Vectored Vaccine: The Janssen (Johnson
& Johnson) SARS-CoV-2 vaccine Ad26 platform encodes a stabilized variant of the SARS-CoV-
2 S protein. The vaccine is administered as a one-dose intramuscular injection. The briefing
document supplied to the FDA for EUA approval revealed five common systemic side effects
(SEs) to the vaccine and three localized SEs (Janssen, 2021). The Phase 3 randomized and
placebo-controlled trial revealed both categories of SEs were reported by more than 50% of the
18-59 years of age participants (systemic SEs 61.5%, local SEs 59.8%). The major concern for
aerospace safety of flight is the systemic SEs (fatigue, headache, myalgia, nausea, and fever)
with median time to onset of two days and a median duration of two days. Local SEs would
become an aerospace safety of flight concern if they become severe enough to require use of
medication for pain control or they impair daily activities or flight egress. Local SEs of this
severity were only reported by 0.9% of trial participants, 18-59 years of age. For personnel in a
flight duty status (all classes) who receive the Janssen vaccine, a “self-limited” grounding period
of 48-hours is recommended to assess for onset of systemic SEs. The development or
presence of any systemic SE during these 48-hours requires extending the “self-limited”
grounding period for a total of 72-hours, regardless of when SEs resolve, to allow for full
recovery. If systemic SEs persist for greater than 72-hours, any personnel in a flight duty status
should see their Aerospace Medicine provider for evaluation. Rare cases of Guillain-Barré
Syndrome and thrombosis with thrombocytopenia, most commonly affecting young women,
have also been reported following administration of this vaccine, but a causal link has not been
established. If any health problems develop besides known side effects, personnel must also
see their Aerospace Medicine provider for evaluation. Recipients of Janssen COVID-19 Vaccine

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 13
should be instructed to seek immediate medical attention if they develop shortness of breath,
chest pain, leg swelling, persistent abdominal pain, neurological symptoms (including severe or
persistent headaches or blurred vision), or petechiae beyond the site of vaccination. The
presence and severity of symptoms may require the grounding of some personnel for greater
than 72-hours. Medical evaluations for return to flight status in these prolonged symptomatic
cases shall follow the return to flight status symptomatic protocols outlined in ALNAV 096/20.
Aerospace Medicine providers administering the vaccine should review further information
provided by the FDA (U.S. Food & Drug Administration, 2021).
Adverse Events and Side Effects of CoVID-19 Vaccines:
The spike protein region of the SARS-CoV2 virus appears to be the most highly correlated with
systemic toxicity occurring in the small percentage of individuals which suffer highly
symptomatic CoVID-19 infections, usually in the geriatric population or in individuals with co-
morbidities. Principle clinical sequelae include hemagglutination, leading to small vessel
thrombi, and auto-immune reactions that can manifest as cytokine release syndrome. Since the
mRNA in the vaccine codes for an almost identical copy of the SARS-CoV2 spike protein,
similar effects can be observed in the immunized, after the spike protein is translated and
expressed on cell surfaces (Kanduc and Shoenfeld, 2020; Charlie-Silva, et al, 2021). The
adverse events (AE), possibly related to spike protein expression in vascular endothelial cells,
can be placed into three principle categories:
1) Auto-immune - chiefly manifest by conditions such as diffuse dermatitis or
myocarditis.
2) Neurologic - manifested by conditions such as transverse myelitis, Guillain-Barre
syndrome or Bell’s Palsy.
3) Provoked clotting cascade / platelet aggregation - manifested by conditions such as
pulmonary embolus and stroke.
Per the CDC VAERS database, as of this revision, adverse events are reported in
approximately 0.34% (~450K/130m), severe AE’s in 0.027% (~35K/130m) and deaths in
0.0077% (~10K/130m) of those immunized with U.S. FDA EUA-approved CoVID-19 vaccines.
This information will fluctuate as adverse events are continually reported into the database. One
Harvard study estimates that only 1% of all vaccine-related AE’s are posted into the CDC
VAERS reporting system, so the actual rates of side effects may be significantly higher
(Lazarus, et al, 2011, and Davenport, 2000). Higher rates of AE’s, serious AE’s and deaths
have been observed in the United Kingdom (MHRA Yellow Card System) and European
(EudraVigilance) reporting platforms, but this may be due to inclusion of the Astra-Zeneca
(Vaxzevria™) COVID-19 Vaccine (ChAdOx1-S [recombinant]) immunization data, which has
shown an increased rate of thrombotic events after injection, and is not approved for use by the
FDA in the U.S via EUA.
References:
(1) Pfizer-BioNTech. (2020, Dec 10). Vaccines and Related Biological Products Advisory
Committee Meeting (FDA Briefing Document)
(2) Moderna. (2020, December 17). Vaccines and Related Biological Products Advisory
Committee Meeting (FDA Briefing Document)
(3) Cohn, A and Mbaeyi, S (2020, Dec 13) What Clinicians Need to Know About the Pfizer
BioNTech COVID 19 Vaccine. Centers for Disease Control and Prevention
https://www.cdc.gov/vaccines/covid-19/downloads/pfizer-biontech-vaccine-what-Clinicians-
need-to-know.pdf
(4) U.S. Food & Drug Administration. (2021, Jul 7). Moderna COVID-19 Vaccine

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 14
https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-
covid-19/moderna-covid-19-vaccine
(5) Janssen Biotech, Inc. (2021, Feb 26). Vaccines and Related Biological Products
Advisory Committee Meeting (FDA Briefing Document)
(6) U.S. Food & Drug Administration. (2021, Jul 20). Pfizer-BioNTech COVID-19 Vaccine
https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-
covid-19/pfizer-biontech-covid-19-vaccine
(7) U.S. Food & Drug Administration. (2021, Jul 13). Janssen COVID-19 Vaccine
https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-
covid-19/janssen-covid-19-vaccine
(8) Kanduc, D, Shoenfeld, Y. Molecular mimicry between SARS-CoV-2 spike glycoprotein
and mammalian proteomes: implications for the vaccine. Immunologic Research (2020)
68:310–313 https://doi.org/10.1007/s12026-020-09152-6
(9) Charlie-Silva, I, et al. An insight into neurotoxic and toxicity of spike fragments SARS-
CoV-2 by exposure environment: A threat to aquatic health.
https://doi.org/10.1101/2021.01.11.425914
(10) Vaccine Adverse Event Reporting System, Center for Disease Control website
https://www.cdc.gov/vaccinesafety/ensuringsafety/monitoring/vaers/index.html
(11) Lazarus, R., et al. Electronic Support for Public Health- Vaccine Adverse Event
Reporting System report to Agency for Healthcare Research and Quality (AHRQ) U.S.
Department of Health and Human Services. 9/30/2010
https://digital.ahrq.gov/sites/default/files/docs/publication/r18hs017045-lazarus-final-report-
2011.pdf
(12) Davenport, K., Vaccines and The National Vaccine Injury Compensation Program
(NVICP) (2000 Paper) Digital Access to Scholarship and Harvard, Harvard Library Vaccines and
The National Vaccine Injury Compensation Program (harvard.edu)
(13) European Medicines Agency (EudraVigilance Adverse Events webpage)
https://www.ema.europa.eu/en; https://www.ema.europa.eu/en/human-
regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-
vaccines/covid-19-vaccines; https://www.ema.europa.eu/en/human-regulatory/research-
development/pharmacovigilance/eudravigilance/eudravigilance-electronic-reporting
(14) Medicines and Healthcare products Regulatory Agency (MHRA) of the United Kingdom
webpage https://yellowcard.mhra.gov.uk/; Official MHRA side effect and adverse incident
reporting site for coronavirus treatments and vaccines | Coronavirus (COVID-19)
SMALLPOX
BACKGROUND: The World Health Organization effectively used smallpox vaccine to eradicate
natural smallpox from the planet however regimes hostile to the United States may possess
strains of the smallpox virus for use as a biological weapon. While routine vaccination is not
recommended for the general population, military and other personnel who serve in high risk
parts of the world may receive smallpox vaccine to protect them from the disease in the event of
a biological attack.
Expect more side effects within the vaccinated population than normally seen with other
vaccines. One expert stated that approximately 10% of vaccine recipients may have side effects
significant enough to cause possible distraction during flying activities. The time range for
development of side effects varies from day 0 until day 14, with most occurring within 3 to7 days
post-vaccination.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 15
Grounding Period: In view of the complications seen with the smallpox vaccination, a 24
hour grounding period is required. It is recognized that complications from the immunization
are most likely in the 3 to 7 day period post immunization. For this reason, close observation
and follow-up is recommended by the Designated Aeromedical personnel or health care
provider. Personnel should be specifically briefed to report any symptoms or complications
during this 3 to 7 day period and to have them evaluated. Depending on the severity, the Flight
Surgeon (or Aeromedical Examiner or Aeromedical Physician Assistant) may ground the aviator
until symptoms have resolved.
ADDITIONAL INFORMATION: Please review the attached "Smallpox Fact Sheet - Information
for Clinicians" and visit the CDC web site and military smallpox website
(http://www.vaccines.mil/) for additional information. Use the CDC Smallpox Adverse Event
Reporting web site to report any adverse events resulting from the administration of the
smallpox vaccination.
TYPHOID AND ORAL TYPHOID:
Vaccine is made of a killed suspension of the bacteria, or a new oral 4 dose preparation. The
injection is a 0.5 cc IM dose at zero and four weeks with about 50-76% efficacy, and protection
for travelers to endemic areas lasts only a few months. This is contrasted with the oral form,
which is equally efficacious but confers immunity to the 21a strain that lasts for years (booster
required at least every 4 years). It is given every other day before meals for a total of 4 doses,
and must be kept refrigerated. Errors in compliance reached 30% of individuals in one study, so
direct observation may be the way to go. Adverse reactions to typhoid injections include
frequent fever, local swelling and pain, and consequently require a 12 hour downing period.
There are no reactions reported to the oral typhoid, therefore no grounding is necessary.
YELLOW FEVER:
This vaccine is used to prevent infection with this flavivirus and its subsequent jaundice,
hemorrhage, and albuminuria in travelers to endemic areas (e.g. South America and Africa). It is
given as a 0.5 cc SC dose. Booster vaccinations are recommended every 10 years. Efficacy is
noted to be high, but adverse side effects include encephalitis/encephalopathy (though fewer
than one in a million cases), and anaphylaxis in those individuals allergic to eggs. A 12 hour
grounding period is recommended for this vaccination.
COMBINED ADMINISTRATION OF VACCINATIONS:
A number of these vaccines can be given together. Generally, any live virus vaccine can be
given with any killed agent or toxoid as long as they are given at the same time and in different
anatomic locations. For example, typhoid may be given with either plague or yellow fever.
Hepatitis A and yellow fever may be given in the same session. One exception to this is cholera
and yellow fever. Administration of these vaccines within 3 weeks of one another results in a
poor antibody response. Unless there is insufficient time, 3 to 4 weeks between live virus
vaccinations should be sought for maximal antibody production. If possible, vaccines frequently
associated with systemic side effects (cholera, typhoid and plague) should not be given
simultaneously so that toxicities will not overlap and that a causative agent can be determined
should a reaction occur.
PREGNANCY AND VACCINATIONS:

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 16
Refer to specific immunization guidelines for vaccination recommendations and precautions
during pregnancy.
GROUNDING FOR VACCINATIONS:
OPNAVINST 3710.7 series requires a 12 hour grounding period following immunizations unless
otherwise specified in this document. The specific guidelines and grounding periods for each
vaccination are described above. As per MANMED Article 15-77, the administration of routine
immunizations that require a temporary grounding, do not require issuance of an Aeromedical
Grounding Notice. This is a “self-limited” grounding period allowed in the absence of adverse
side effects.

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 17
18.6 MISCELLANEOUS MEDICATIONS
Last Reviewed: March 22 Last Revised: March 22
ALLOPURINOL: CD. Waivers are recommended to SG3, Class II, or Class III. Re-evaluation
for upgrade from SG3 to SG1 is considered in 3 months if member remains
asymptomatic and on a stable dose of medication.
ANTIHISTAMINES (SEDATING):
CD. The member should be grounded for the duration of therapy.
ANTIHISTAMINES (NON-SEDATING):
NCD. Allegra and Claritin are NCD if given in accordance with the Allergic/Vasomotor
Rhinitis section of the Waiver Guide. Refer to this section for additional restrictions and
clarification. Zyrtec, although considered by some to be non-sedating, still has a
moderate sedating effect and is therefore not approved (CD) for use in aviation
personnel.
CLOMIPHENE (CLOMID): CD- No Waivers.
CONTRACEPTIVES:
DEPO-PROVERA : NCD. Any grounding period at discretion of the local Flight Surgeon
to assure tolerance.
LEVONORGESTEROL (NORPLANT): NCD. Any grounding period at discretion of the
local Flight Surgeon to assure tolerance.
PROGESTASERT IUD: NCD. Any grounding period at discretion of the local Flight
Surgeon to assure tolerance.
DECONGESTANTS: CD. Requires temporary grounding while in use.
FINASTERIDE (PROPECIA/PROSCAR):
CD. A waiver can be considered after a two week grounding. If the patient remains
asymptomatic, a LBFS may issue an up chit. Finasteride may be utilized for prostatic
hypertrophy or alopecia. DoD pharmacy does not allow prescriptions of finasteride for
hair loss.
H2 BLOCKERS:
RANITIDINE, CIMETIDINE, FAMOTIDINE: CD. A waiver is required for any chronic use.
Refer to the Gastroenterology Waiver Guide section on reflux esophagitis for additional
information.
INHALED STEROIDS: CD. Decisions are individualized. Any chronic use requires a waiver.
Call NAMI Code 53HN for additional guidance.
ISOTRETINOIN (ACCUTANE, AMNESTEEM, CLARAVIS, SOTRET): CD. No waiver.
Resumption of flight status is permitted after member is off medication for 3 months, has
a normal slit lamp exam, and triglyceride levels are documented as normal. Cystic acne,
if severe enough to need Accutane, may be disqualifying. The 3 month delay after

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 18
cessation of treatment also allows for an evaluation of how the member does without the
medication.
LEVOTHYROXINE (SYNTHROID): CD. A waiver may be requested when member is clinically
and chemically euthyroid on stable dosage.
LINDANE (KWELL): NCD. Requires a 48-hour grounding period. Kwell can be absorbed in
variable amounts and give some significant CNS side effects. Aviation personnel must
be grounded for 48 hours after the compound is washed off.
MESALAMINE (ASACOL, ROWASA, ETC.): CD. A major advantage of mesalamine is that it
avoids some side effects associated with the sulfapyridine moiety of sulfasalazine.
Waiver will be considered after maintaining clinical remission for one month without
evidence of side effects.
MINOXIDIL (TOPICAL): Use of topical minoxidil (2% or 5%) changed to not considered
disqualifying for SG 1, SG2, SG3, Class II, Class ID, and Class IV (all types). First time use of
topical minoxidil (2% or 5%) results in a three day mandatory grounding period in order to
observe for any untoward effects. This grounding period does not need to be monitored by the
flight surgeon. With symptoms of contact dermatitis that would be detrimental to wearing
required flight gear, the member shall discontinue use of the medication. With any symptoms of
hypotension, the member shall discontinue use of the medication and report to the flight
surgeon.
NEDOCROMIL (TILADE): CD. Tilade may be considered for waiver for in designated aviation
personnel for the preventive treatment of mild to moderate asthma or cold-induced and
exercise-induced bronchospasm. Member will be eligible for waiver consideration and
return to flight status at a minimum two weeks after remaining symptom free on a stable
dose of medication with demonstrated normal pulmonary function tests. Waivers are
restricted to non-high performance aircraft.
NASAL STEROIDS:
FLONASE, NASONEX, RHINOCORT: NCD. Refer to the Allergic/Vasomotor Rhinitis
section of the Waiver Guide under Ear/Nose/Throat for additional restrictions and
clarification.
SMOKING CESSATION:
NICORETTE GUM: NCD if the following conditions are met:
1. Enrolled in formal organized stop smoking program.
2. Close observation by flight surgeon.
3. No adverse effects.
4. Duration of use does not exceed three months.
NICOTINE TRANSDERMAL SYSTEM (NICODERM): NCD. Aviators should be grounded for 48
hours following application of first patch.
All other medications for tobacco cessation are not approved for use by personnel on
active flight status, so require grounding during treatment. This can often be planned to
U.S. Navy Aeromedical Reference and Waiver Guide Medications - 19
coincide with non-flying periods. Guidance for timing of return to flight is based on the
elimination half-life of the drug being used, as follows:
VARENICLINE (CHANTIX
®
): Varenicline has an elimination half-life of 24 hours, so
individuals should be grounded for one more week after finishing Chantix
BUPROPION (ZYBAN
®
, WELLBUTRIN
®
): Bupropion is cleared more quickly, but only
about 1% is excreted unchanged in the urine; the rest is metabolized to three major active
metabolites, threohydrobupropion, erythrohydrobupropion and hydroxybupropion, which
accumulate to levels much higher than the parent compound and can have extended half-lives
of as long as 43 hours. Individuals taking bupropion should therefore be kept down for two
weeks following completion of treatment.
NON-STEROIDAL ANTI-INFLAMMATORY DRUGS (NSAIDS):
ASPIRIN: NCD for occasional analgesic use or at cardioprotective dosing. Other chronic
use is CD and requires a waiver.
IBUPROFEN/NAPROXEN: NCD. Medication can be used for short term use under
direct supervision of Flight Surgeon. Any chronic or high dose use is disqualifying. If
recommending that an aviator continue to fly during treatment, consider the underlying
reason for its use. It may be the condition which is disqualifying.
INDOCIN: CD. No waiver. Ground during medication use and for two weeks after
medication is completed.
PHOSPHODIESTERASE INHIBITORS (PDI):
SILDENAFIL (VIAGRA), VARDENAFIL (LEVITRA), TADALAFIL (CIALIS):
1. Long-acting PDI, tadalafil (Cialis): CD. No Waivers
2. Short-acting PDI: sildenafil (Viagra), vardenafil (Levitra): CD. Waivers possible for:
a. Initial Applicants – considered on a case-by-case basis
b. Designated Personnel – may request waiver after evaluation for the cause of ED.
3. Information Required:
a. AMS – history, physical, lab, prior treatment course, side effect.
b. Consultation – Urology, Internal or Family Medicine – Must evaluate causes of ED
including co-morbid conditions such as cardiovascular (hypertension,
atherosclerosis, or hyperlipidemia), diabetes mellitus, depression and alcoholism
c. An exercise stress test should be completed prior to waiver submission whenever
indicated. If test is not performed, reasons should be substantiated in AMS or by
consultants.
4. Follow-up: annual – to assess efficacy, side effects, and significant changes in health
status including medications
5. Treatment:
a. Must be free of side-effect for 2 doses after beginning medication before returning to
flying duty
b. No flying duties within 12 hours of last dose (medicine use to briefing time)

U.S. Navy Aeromedical Reference and Waiver Guide Medications - 20
6. Links:
a. Diagnostic evaluation: Link to diagnostic evaluation
(http://www.aafp.org/afp/2000/0101/p95.html)
b. Precautions: Link to precautions (http://www.aafp.org/afp/1999/0915/p1159.html)
c. Medications: sildenafil (Viagra®) , vardenafil (Levitra®)
PROBENECID:
CD. Waiver is required for any long term treatment.
PROTON PUMP INHIBITORS:
OMEPRAZOLE, LANSOPRAZOLE, RABEPRAZOLE: CD. Waiver required for chronic
use. Refer to the Gastroenterology Waiver Guide section on reflux esophagitis for
additional information.
SUCRALFATE (CARAFATE):
NCD when used in dosages of 1 gm bid or less. However, the diagnosis of peptic ulcer
disease is certainly CD and requires a waiver.
SULFASALAZINE (AZULFIDINE):
CD. Waiver considered after maintaining clinical remission for one month without
evidence of side effects.
TAMOXIFEN: CD. No Waiver.
TOPICAL COMPOUNDS:
As a general rule, medications applied to the surface of the body which are not absorbed
to any significant extent are NCD. However, please see notes on Kwell. The
recommended initial treatment is over a weekend to allow return to flight duties the
following Monday, thus minimizing flight schedule loss.
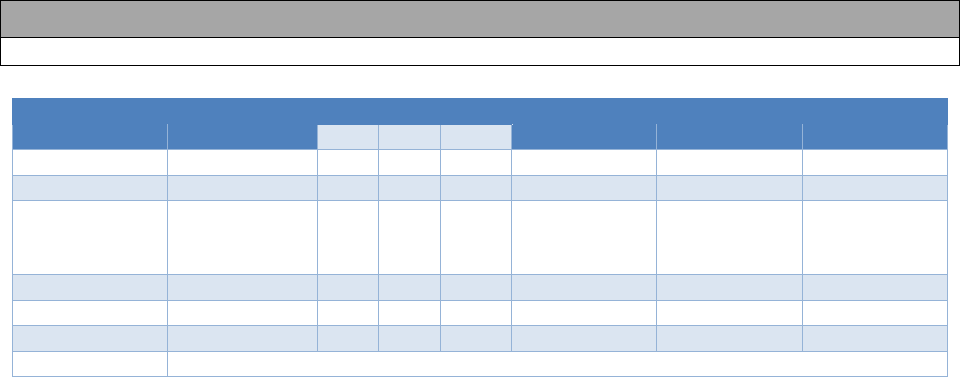
U.S. Navy Aeromedical Reference and Waiver Guide Medications - 21
18.7 PSYCHOTROPIC MEDICATIONS
Last Reviewed: October 18 Last Revised: October 18
Applicant
Class I
Class II
Class III
Class IV
SG1
SG2
SG3
CD
X
X
X
X
X
X
X
NCD
WR
case-
by-
case
case-by-case
case-by-case
case-by-case
WNR
X
X
X
LBFS
No
No
No
No
No
No
No
EXCEPTIONS
LIMDU/ PEB
May be required, depending on the severity of the clinical condition.
AEROMEDICAL CONCERNS: Untreated or poorly-controlled psychiatric symptoms can
negatively impact occupational functioning, as well as quality of life and social, emotional, and
cognitive functioning of affected individuals. A 2002 study found that when advised of the FAA’s
policy at the time (that each pilot would be grounded until the depression had cleared and the
medication had been discontinued for approximately 3 months) the pilots indicated their intended
responses to the prospect of not flying for 9 months or more. Of the 1200 pilots surveyed, some
59% (710) said they would refuse the medication and continue to fly. About 15% (180) indicated
an intention to take the medication and continue their flight duties without informing the FAA. The
remaining 25% (300) said they would take sick leave, undergo the recommended treatment, and
return to work when aeromedically cleared to do so.
1
In addition to concerns about the avoidance
of treatment for clinically and aeromedically significant psychiatric symptoms, and the unmanaged
risks associated with treatments not disclosed to the aviator’s flight surgeon, premature
discontinuation of a psychotropic medication, which may be driven by a desire or operational need
to return to Duty Involving Flying (DIF), may result in a return of psychiatric symptoms, worsen
the overall course of the disease process, and necessitate grounding. For these reasons, waivers
for return to DIF will be considered according to the following guidelines.
INITIAL WAIVER PROCESS (WAIVERS WILL NOT BE CONSIDERED FOR APPLICANTS):
Designated Flying Class I, SG3 aviators and Flying Class II-IV service members on aviation status
are eligible for waiver consideration for continuous (i.e., not PRN) pharmacotherapy with FDA-
approved psychotropic medication(s) for the conditions listed below:
1. Depressive Disorders (ARWG Section 14.4)
2. Anxiety Disorders (ARWG Section 14.5)
3. Obsessive-Compulsive Disorder and Related Disorders (ARWG Section 14.6)
4. Trauma and Stressor-Related Disorders (ARWG Section 14.7)
Waiver consideration may be requested after a suitable Period of Observation in a Non-flying
Status (PONS) has elapsed (Waivers will not be considered for APPLICANTS). The PONS
begins once the psychiatrist* has declared, by way of a formal medical record entry, that the
service member’s condition is in full remission. The PONS is dictated by the psychiatric diagnosis
and is outlined in the relevant section of the ARWG. During the PONS and thereafter:
1. The dose of the medication(s) must remain stable.
2. There must be no aeromedically-significant medication side effects.
U.S. Navy Aeromedical Reference and Waiver Guide Medications - 22
3. The clinical condition must remain in stable remission.
INFORMATION REQUIRED FOR INITIAL MEDICATION WAIVER:
1. The psychiatric condition has to be waiverable.
2. All waiver requirements for that psychiatric condition must be met. Without this, no request
for a medication waiver can be considered.
Note: Medication use and waiver provisions may be included in the comprehensive AMS. (Only one AMS is required.)
3. A comprehensive NAMI Psychiatric and Neuropsychological assessment is required as
part of any waiver request.
INFORMATION REQUIRED FOR MEDICATION WAIVER CONTINUANCE:
1. A comprehensive local psychiatric* evaluation must be conducted every 6 months.
2. An Aeromedical Summary (AMS) documenting the presence or absence of interim
symptoms must be submitted to NAMI (via AERO) annually. This summary must include
a copy of the comprehensive local psychiatric* evaluation reports.
3. CogScreen-AE testing must be performed annually and the results must be submitted via
AERO.
INFORMATION REQUIRED FOR CHANGES IN MEDICATION DOSING (Including dose
increase, decrease, or discontinuation):
1. If a medication is discontinued or the dose is decreased, a NAMI psychiatric evaluation is
required. This will be conducted no earlier than 60 days after medication reduction or
discontinuation.
2. Local mental health evaluations are required 6 and 12 months after medication
discontinuation. The results of these evaluations are to be submitted with the next annual
flight physical via AERO.
3. If the dose of a previously waived medication is increased, please contact NAMI
Psychiatry for guidance.
Notes:
For the purpose of standardization of terms used in discussing aeromedical disposition, the
following definitions will be used. These are based on recommendations of a task force convened
in 1988 to create a consensus vocabulary for discussing Major Depressive Disorder.
2
Partial Remission is a period during which an improvement of sufficient magnitude is observed
that the individual is no longer fully symptomatic (i.e. no longer meets full syndromal diagnostic
criteria for the disorder) but continues to suffer more than minimal symptoms.
Response is the point at which a partial remission begins (in pharmaceutical studies, medication
response is typically defined as a 50 per cent reduction on a symptom severity measure, such as
the Hamilton Depression Rating Scale).
Full Remission is a relatively brief period (DSM-5 suggests 2 months for Major Depressive
Disorder) with no symptoms, or “only one or two symptoms to no more than a mild degree.” While
U.S. Navy Aeromedical Reference and Waiver Guide Medications - 23
“mild” symptoms may be reasonable in general clinical settings, the standard for aviation duty
must be higher, so in this context “no symptoms” is the standard.
Recovery is remission sustained for a minimum specified period of time. For the purpose of
waiver consideration, refer to the relevant section of the ARWG for the minimum time to recovery
for a particular condition. Recovery can occur in response to treatment, but can also happen
spontaneously in the natural course of the condition.
Relapse is defined as a return of symptoms satisfying the full syndromal diagnostic criteria for an
episode that occurs during the period of remission, but before recovery as defined above.
Relapse can represent a change from either partial or full remission to fully syndromal diagnostic
criteria for the disorder. The reason to distinguish between a relapse and a recurrence is the idea
that relapse represents the return of the symptoms of a still ongoing but symptomatically
suppressed episode, while a recurrence represents an entirely new episode, with significantly
different prognostic implications.
Recurrence is the appearance of a new episode and so can only occur during a recovery.
*If a psychiatrist is not available, please contact NAMI Psychiatry at (850) 452-2783 or
1
Hudson DE Jr. SSRI use in professional aircrew. Panel presentation. Aerospace Medical
Association annual meeting. 9 May 2002, Montreal, Canada.
2
Frank E, Prien RF, Jarrett RB, Keller MB, Kupfer DJ, Lavori PW, Rush AJ, Weissman MM:
Conceptualization and rationale for consensus definitions of terms in major depressive disorder.
Arch Gen Psychiatry 1991; 48:851-855.
